 In general the higher the energy of the state that undergoes nuclear fission the more likely a symmetric fission is, hence as the neutron energy increases and/or the energy of the fissile atom increases the valley between the two peaks becomes more shallow, for instance the curve of yield against mass for Pu-239 has a more shallow valley than that observed for U-235 when the neutrons are thermal neutrons. In this bar chart results are shown for different cooling times (time after fission).īecause of the stability of nuclei with even numbers of protons and/or neutrons the curve of yield against element is not a smooth curve. Note that in the calculations used to make this graph the activation of fission products was ignored and the fission was assumed to occur in a single moment rather than a length of time. Z - This is a typical distribution for the fission of uranium. 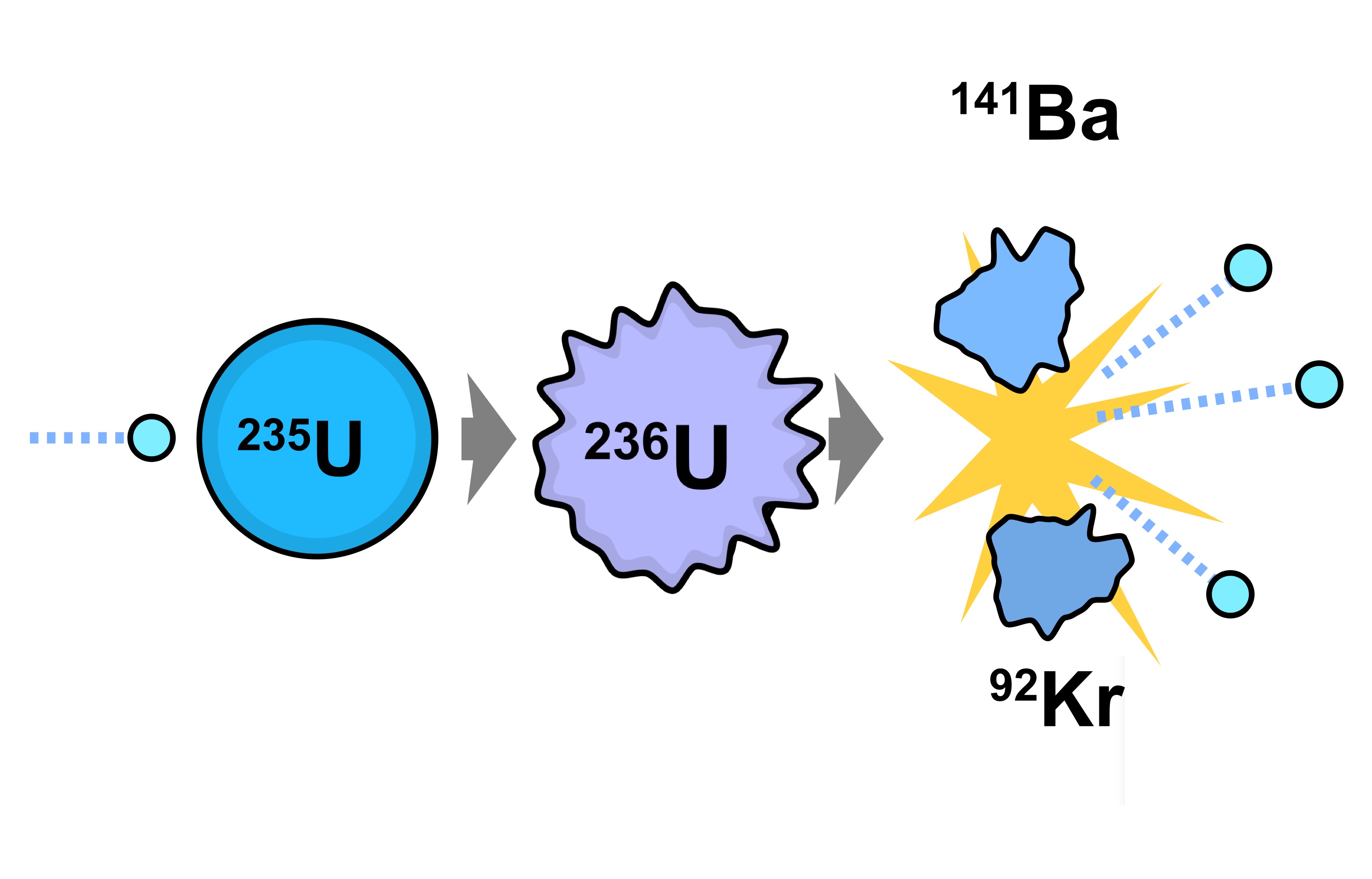
This is due to the fact that the fission event causes the nucleus to split in an asymmetric manner. If a graph of the mass or mole yield of fission products against the atomic mass of the fragments is drawn then it has two peaks, one in the area zirconium through to palladium and one at Xenon through to neodymium. A more detailed description of individual products, organized by element, may be found here Mass vs. After a few years, the radiation is dominated by Sr-90 and Cs-137, whereas in the period between 10,000 and a million years it is Technetium-99 that dominates. After two to three years, Ce-144/ Pr-144, Ru-106/ Rh-106, and Pm-147 are the bulk of the radioactivity. After about four months Ce-141, Zr-95/ Nb-95, and Sr-89 represent the largest share of radioactive material. One peak gives a peak (expressed by atomic number) at about strontium to ruthenium while the other peak is at about tellurium to neodymiumĪccording to Jiri Hala's textbook the radioactivity in the fission product mixture (due to an atom bomb) is mostly caused by short-lived isotopes such as I-131 and Ba-140. The majority of the mass yield of the fission products occurs in two peaks. The fission products include every element from zinc through to the mid to late lanthanides. This is the source of delayed neutrons which play an important role in control of a nuclear reactor. Sn-126 (even higher energy gamma, but long halflife of 230,000 years means a slow rate of radiation release, and the yield of this nuclide per fission is very low)Ī few neutron-rich and short-lived initial fission products first decay by emitting a neutron.Cs-137 (high energy gamma, halflife 30 years).Sr-90 (high energy beta, halflife 30 years).Many of the early decays of short-lived nuclei are high-energy, but the beta and gamma emissions of most of the long-halflife fission product nuclides are of relatively low energy and therefore of relatively less biological risk. However, fission products do not emit alpha particles only the heavy actinide nuclei produced by neutron absorption without fission do that. Some beta decays and isomeric transitions are accompanied by gamma radiation. The first beta decays are rapid, but in some fission products, the last one or two decays have a long halflife, so that the fission product which has not yet undergone these last decays is a nuclear waste that must be stored safely for a long time. stable 56% ruthenium-100 is 56% neutrons, stable xenon-134 is 60%), and therefore undergo beta decay towards stable nuclei, converting a neutron to a proton with each beta emission. 61% of the nucleons in uranium-235 are neutrons), the initial fission products are almost always more neutron-rich than stable nuclei of the same mass as the fission product (e.g. Since the heavy nuclei that can undergo fission are particularly neutron-rich (e.g. This is because some of the mass is lost as free neutrons and large amounts of energy. The sum of the atomic weight of the two atoms produced by the fission of one atom is always less than the atomic weight of the original atom. 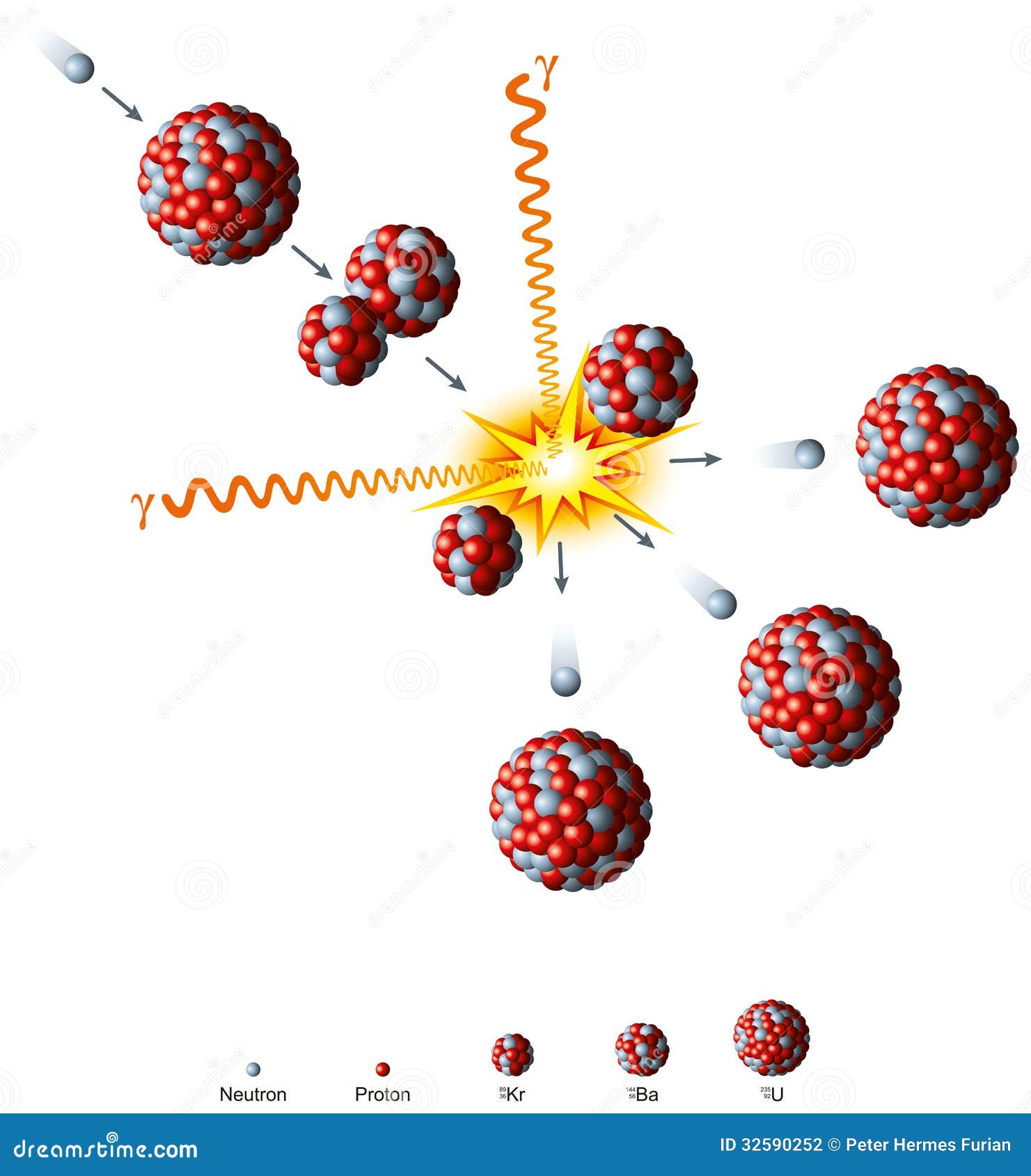
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
